
Bacteria Under the Microscope
What Are Bacteria? Bacteria are single-celled organisms that are defined as prokaryotes, these are organisms that have cells with no defined nucleus or other specialized organelles. In total, there are

Compound Microscopes vs. Stereo Microscopes
Compound Microscopes and Stereo Microscopes: Similar but Different Stereo microscopes and compound microscopes are two distinct types of light microscopes that have many similarities, but some fundamental differences. It’s certainly

Different Types of Laboratory Microscopes and Their Functions
Types of Laboratory Microscopes There are many different types of microscopes used in modern pathology laboratories and research departments around the world. These typically include stereo, compound, digital and pocket
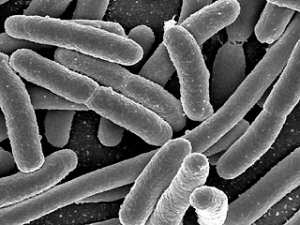
Disadvantages of Electron Microscopes
What are Electron Microscopes? Electron microscopes (EM’s) are very sophisticated and powerful pieces of equipment that have revolutionized the world of science and medicine. Thanks to the EM for the

Easy and Fun Microscope Experiments
Fun and Safe Microscope Experiments for Beginners Although a microscope is well associated with serious and important activities performed by medical professionals and scientists, it can also be used for

How to Clean Your Microscope
Keep Your Microscope Clean Microscopes are highly sophisticated and often expensive pieces of equipment that have the potential to last many years. However, even though the structure of microscopes is

Microscopy and Related Techniques
What is Microscopy? Microscopy is the term used for the practice of using a microscope. The microscope is an essential tool that is widely used by medical professionals, scientists and

The History of Optical Microscopes
Development of Lenses Microscopes are amazing tools that have enabled man to make new scientific discoveries, diagnose and treat human disease, as well as make intricate things that require powerful

Types of Objective Lens & Their Functions
Microscope Lenses Provide Magnification Power Light microscopes are relatively complex pieces of equipment in nature with multiple different parts, some which are more complex than others. The lenses of the

What is an Electron Microscope?
Electron Microscopes Use a Beam of Electrons Instead of Light Light microscopes have many important uses, especially for medical professionals and researchers who use them to help make a diagnosis
